Water Heater Anode Rods
In a water heater, an anode rod, also referred to as “sacrificial” rod, is consumed as it “gives up” its electrons to protect the carbon steel tank (cathode). The two main factors that contribute to anode activity are anode material, and water chemistry.
The Anode:
Magnesium is a leading anode material given its ability to best protect the steel tank. It “gives up” electrons more freely than aluminum. In some situations where water is more conductive, i.e. softened water or lower pH levels, this sacrificial process is accelerated; in those cases, aluminum is a better choice.
Water Chemistry:
Water chemistry not only affects conductivity and how quickly the anode is consumed, but also contributes to situations like “rotten egg smell.”
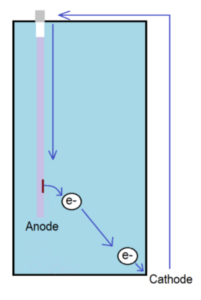
Water hardness is often considered, but other conditions affecting anode rod performance include:
- Dissolved solids like calcium & lime.
- Sulfur content.
- Use of a water softener.
- Municipal vs. well water.
- Non-harmful sulfur eating bacteria.
Some of these conditions could be defined as “good” by conventional water testing standards, while still accelerating the sacrificial process of the anode.
Indications that water testing and/or anode material change may be needed include, but are not limited to:
- “Air” or “sputtering” in the hot water when faucets are turned on.
- Smelly hot water.
- Discolored or cloudy hot water.
Service Recommendations
For air in the hot water lines, service action includes:
- Replace the anode rod(s) with a less reactive material (magnesium to aluminum, or aluminum to aluminum alloy)
- Water chemistry frequently changes, be sure to understand current water conditions. Water test parameters to consider:
- pH level (7 is neutral and ideal).
- Hardness check:
- Overly softened water will increase conductivity.
- 10 – 12 grains per gallon hardness is a common target.
With smells or discoloration in the water, BWC recommends draining the hot water system and chlorinating the water heater and hot water piping. See Service Bulletin 109 – Hydrogen Sulfide Odor and Chlorinating Water Heaters.

 USA
USA CAN
CAN

